100 FAQs on Newer Biomarkers in Acute Coronary Syndrome
100 FAQs on Newer Biomarkers in Acute Coronary Syndrome
HIGH-LEVEL, NEET-SS / DM–oriented set of 100 FAQs on Newer Biomarkers in Acute Coronary Syndrome, written with conceptual depth, exam traps, discriminator points, and “why not the others” logic.
This is not undergraduate material. It is aligned to recent trials, universal MI definitions, and viva-level questioning.
SECTION A: FOUNDATIONAL CONCEPTS (Q1–Q15)
1. What fundamentally differentiates a “biomarker of myocardial injury” from a “biomarker of myocardial infarction”?
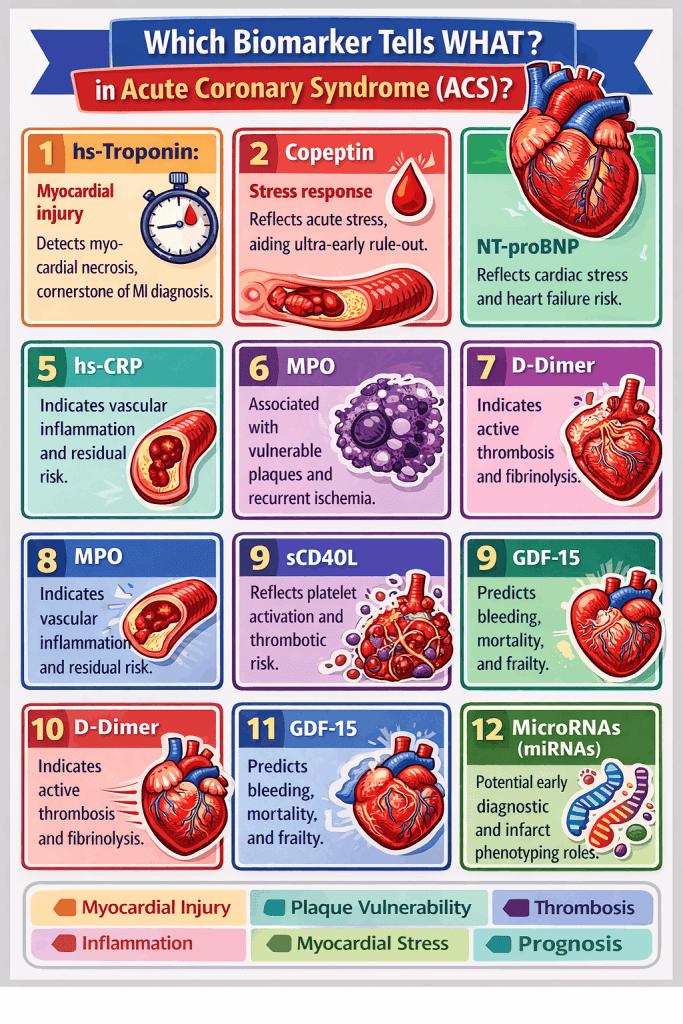
A biomarker of injury (e.g., hs-troponin) indicates cardiomyocyte injury irrespective of mechanism, whereas myocardial infarction requires ischemia + injury + clinical context.
Extra point: This distinction underpins the Universal Definition of MI and explains troponin elevation in sepsis, CKD, and PE.
2. Why is hs-troponin considered a “gatekeeper biomarker” in ACS?
Because it is mandatory for diagnosis, risk stratification, and management decisions.
Exam pearl: No newer biomarker has displaced troponin in guideline algorithms.
3. What is meant by “delta troponin,” and why is it critical?
The change in troponin over time (absolute or relative) distinguishes acute injury from chronic elevation.
Trap: A single elevated troponin ≠ MI.
4. Why did CK-MB lose relevance despite myocardial specificity?
Lower sensitivity, delayed rise, and inferior prognostic power compared to hs-troponin.
NEET-SS angle: CK-MB is now mainly historical or for reinfarction in select settings.
5. What does “analytical sensitivity” mean in hs-troponin assays?
Ability to measure troponin in >50% of healthy individuals.
Extra point: This enabled 0/1-hour and 0/2-hour rule-out algorithms.
6. Why are sex-specific troponin cutoffs important?
Women have lower 99th percentile values; ignoring this leads to underdiagnosis.
Exam trap: Using a single cutoff disadvantages women.
7. Why is troponin elevation prognostic even when non-ischemic?
It reflects myocardial vulnerability and systemic illness severity.
Key concept: Troponin is a “barometer of risk,” not just infarction.
8. What is the biggest clinical danger of over-interpreting hs-troponin?
Overdiagnosis of MI → unnecessary anticoagulation, angiography, bleeding risk.
9. Can troponin ever be normal in true ACS?
Yes, very early presentation or unstable angina without necrosis.
Pearl: Biomarkers do not replace clinical judgment.
10. Why is timing of sampling as important as absolute value?
Because kinetics define acuity.
Exam angle: Early presenters need repeat sampling.
11. What is “troponin leak”? Is it a valid term?
Colloquial, inaccurate term for myocardial injury.
NEET-SS tip: Avoid in exams and viva.
12. Why are different hs-troponin assays not interchangeable?
Different antibodies, calibration, and cutoffs.
Trap: Use assay-specific reference ranges.
13. Why do guidelines discourage routine use of multiple necrosis markers?
No incremental benefit over hs-troponin.
14. What is the most important limitation of troponin?
Lack of specificity for ischemic etiology.
15. Why has no biomarker replaced troponin despite decades of research?
Because troponin directly reflects cardiomyocyte death with unmatched sensitivity and outcome linkage.
SECTION B: ULTRA-EARLY & STRESS MARKERS (Q16–Q30)
16. What is copeptin biologically?
C-terminal fragment of pre-pro-vasopressin.
Exam pearl: Stable surrogate of vasopressin.
17. Why was copeptin proposed as an ACS biomarker?
Rises immediately with stress, before necrosis occurs.
18. What is the “dual-marker strategy” involving copeptin?
Copeptin + hs-troponin at presentation for rapid rule-out.
Key limitation: Loses value once hs-troponin assays became ultra-sensitive.
19. Why is copeptin NOT widely used today?
Marginal incremental value, poor specificity, added cost.
20. Which conditions falsely elevate copeptin?
Sepsis, stroke, trauma, surgery.
Trap: Stress ≠ ischemia.
21. Can copeptin rule-in MI?
No. Only rule-out adjunct.
22. Why is copeptin strongest in very early presenters (<2 h)?
Troponin may still be negative.
23. What is ischemia-modified albumin (IMA)?
Albumin altered by ischemic oxidative stress.
Exam fact: FDA-cleared but clinically marginal.
24. Why did IMA fail to gain traction?
Poor specificity, influenced by liver disease and systemic illness.
25. Which marker best reflects neurohormonal stress in ACS?
Copeptin.
26. Why are stress biomarkers conceptually appealing but clinically weak?
Stress is ubiquitous in acute illness.
27. Which early biomarker is most influenced by circadian rhythm?
Copeptin.
28. Why do guidelines not recommend copeptin routinely?
No impact on management decisions.
29. Can copeptin guide invasive strategy?
No.
30. NEET-SS takeaway for copeptin?
Understand concept, know limitations, do not overuse.
SECTION C: MYOCARDIAL STRESS & HEART FAILURE RISK (Q31–Q45)
31. What does NT-proBNP reflect in ACS?
Myocardial wall stress and hemodynamic burden.
32. Why is NT-proBNP prognostic but not diagnostic in ACS?
Elevates in HF, AF, CKD, age.
33. Which outcomes does NT-proBNP predict best post-MI?
Heart failure, mortality, arrhythmias.
34. Why can NT-proBNP be elevated with preserved EF?
Diastolic dysfunction and transient ischemic stunning.
35. NT-proBNP vs troponin: key difference?
Troponin = necrosis; NT-proBNP = stress.
36. Which biomarker better predicts sudden cardiac death post-MI?
NT-proBNP.
37. Why is NT-proBNP called a “vulnerable patient marker”?
Integrates cardiac, renal, and systemic risk.
38. What is its biggest confounder?
Renal dysfunction.
39. Can NT-proBNP guide timing of discharge?
Yes, in selected settings.
40. Why is BNP less preferred than NT-proBNP in ACS studies?
Shorter half-life, assay variability.
41. Which biomarker correlates best with infarct size besides troponin?
NT-proBNP.
42. Does NT-proBNP guide revascularization decisions?
Indirectly, via risk stratification.
43. Why is NT-proBNP elevated even in NSTEMI with small infarcts?
Global myocardial stress.
44. Exam trap: NT-proBNP normal excludes MI?
False.
45. NEET-SS summary for NT-proBNP:
Powerful prognostic adjunct, not a diagnostic tool.
SECTION D: INFLAMMATION & PLAQUE VULNERABILITY (Q46–Q65)
46. What does hs-CRP primarily represent in ACS?
Residual inflammatory risk.
47. Why is hs-CRP independent of LDL?
Inflammation and lipids are parallel pathways.
48. Which trials highlighted hs-CRP importance?
JUPITER, CANTOS.
49. Why is hs-CRP prognostic but not diagnostic?
Elevated in many inflammatory states.
50. Which biomarker predicts benefit from anti-inflammatory therapy?
hs-CRP.
51. What is MPO and what does it signify?
Neutrophil activation and oxidative plaque instability.
52. Why is MPO considered a “vulnerable plaque marker”?
Elevates before necrosis.
53. MPO limitation?
Poor specificity and assay variability.
54. What is sCD40L?
Marker of platelet activation and inflammation.
55. Why is sCD40L conceptually attractive?
Links thrombosis and inflammation.
56. Why is sCD40L not used clinically?
Pre-analytical instability, limited validation.
57. What is PAPP-A?
Protease associated with plaque rupture.
58. Why did PAPP-A fail clinically?
Assay interference, poor reproducibility.
59. Which inflammatory marker best predicts recurrent MI?
hs-CRP.
60. Which marker reflects systemic rather than coronary inflammation?
hs-CRP.
61. Can inflammatory biomarkers guide PCI decisions?
No.
62. Why is inflammation a therapeutic target but poor diagnostic tool?
Lack of specificity.
63. Exam trap: Elevated hs-CRP = culprit lesion?
False.
64. Which biomarker explains residual risk after statins?
hs-CRP.
65. NEET-SS takeaway:
Inflammation predicts outcomes, not anatomy.
SECTION E: THROMBOSIS & PROGNOSIS (Q66–Q85)
66. What does D-dimer reflect in ACS?
Active thrombosis and fibrinolysis.
67. Why is D-dimer nonspecific in ACS?
Elevated in PE, cancer, infection.
68. Which ACS phenotype shows highest D-dimer?
STEMI with large thrombus burden.
69. Does D-dimer guide antithrombotic choice?
No.
70. What is GDF-15?
Stress-responsive cytokine.
71. Why is GDF-15 a powerful prognostic marker?
Predicts mortality, bleeding, frailty.
72. Why is GDF-15 poor diagnostically?
Elevated in aging, cancer, CKD.
73. Which biomarker predicts bleeding risk best?
GDF-15.
74. Which score incorporates GDF-15?
ABC bleeding risk score.
75. Why is GDF-15 called a “global risk integrator”?
Reflects cardiovascular and non-cardiovascular risk.
76. Does GDF-15 guide therapy currently?
Limited to research and risk scores.
77. Why is prognostic information alone insufficient for routine use?
Does not change management.
78. Which marker best predicts long-term mortality?
GDF-15.
79. Can D-dimer distinguish Type 1 vs Type 2 MI?
No reliably.
80. Which biomarker predicts frailty post-MI?
GDF-15.
81. Thrombotic biomarkers vs platelet function tests?
Different domains.
82. Why are thrombotic biomarkers underutilized?
Clinical decisions already protocol-driven.
83. Exam trap: Elevated D-dimer mandates anticoagulation?
False.
84. Prognostic vs actionable biomarkers: difference?
Actionable biomarkers change treatment.
85. NEET-SS summary:
Prognosis ≠ diagnosis ≠ therapy.
SECTION F: EMERGING & FUTURE BIOMARKERS (Q86–Q100)
86. What are microRNAs (miRNAs)?
Small non-coding RNAs regulating gene expression.
87. Why are miRNAs exciting in ACS?
Ultra-early release, tissue specificity.
88. Key limitation of miRNAs?
Assay standardization.
89. Can miRNAs differentiate STEMI vs NSTEMI?
Potentially, but not clinically ready.
90. Why are multi-marker panels attractive?
Capture multiple pathophysiologic axes.
91. Why have panels failed clinically?
Complexity without decision impact.
92. What is the “omics” promise in ACS?
Precision phenotyping.
93. Why is cost-effectiveness critical for biomarker adoption?
ACS already resource-intensive.
94. Which biomarker domain has most future potential?
Inflammation-targeted biomarkers.
95. Why will troponin remain central?
Unmatched validation.
96. Can AI replace biomarkers?
Augment, not replace.
97. Biggest mistake in biomarker interpretation?
Ignoring clinical context.
98. How do guidelines view newer biomarkers?
Adjunctive, not primary.
99. What should a NEET-SS candidate remember most?
Know WHAT the marker reflects and WHY it fails.
100. One-line NEET-SS mantra:
“Troponin diagnoses, NT-proBNP stratifies, hs-CRP explains residual risk, others predict but rarely act.”
Best interpretation?
Which imaging finding best supports the diagnosis?
Most accurate diagnosis?
Which biomarker best predicts long-term mortality?
What does MPO elevation indicate?
What clinical risk is most underestimated if GDF-15 is ignored?
Correct classification?
Best explanation?
🧠 ORAL-VIVA RAPID-FIRE CASES
Imaging + Biomarker Discordance (ACS & Mimics)
VIVA 1: Troponin vs CMR
Examiner:
A patient has chest pain, hs-troponin is 8× ULN. CMR shows no LGE. Diagnosis?
Candidate (core answer):
Acute myocardial injury without infarction.
Examiner push:
Why is infarction excluded despite high troponin?
Killer points:
- MI requires irreversible necrosis → LGE
- Troponin reflects injury, not etiology
- Could be type 2 MI, myocarditis early phase, stress injury
Trap:
Do NOT say “false-negative CMR” unless done <24 h
VIVA 2: OCT Trap
Examiner:
NSTEMI patient. OCT shows intact fibrous cap with luminal thrombus. Troponin mildly elevated. What mechanism?
Core answer:
Plaque erosion.
Examiner push:
Which biomarker pattern supports erosion over rupture?
Expected:
- Lower troponin burden
- Less inflammatory biomarkers (lower hs-CRP)
- Often younger, female, smokers
Scoring pearl:
OCT > angiography for mechanism
VIVA 3: IVUS vs Troponin
Examiner:
IVUS shows large plaque burden, positive remodeling. Troponin negative.
Question:
Does this patient have ACS?
Answer:
No myocardial infarction yet, but high-risk vulnerable plaque.
Pushback:
Why troponin negative?
Answer:
No necrosis yet. Imaging precedes biomarkers.
Trap:
Do not equate plaque burden with infarction
VIVA 4: BNP–Troponin Mismatch
Examiner:
ST elevation, modest troponin rise, very high NT-proBNP, echo shows apical ballooning.
Diagnosis?
Takotsubo cardiomyopathy.
Push:
What imaging confirms it?
Answer:
CMR showing edema without LGE.
Examiner trap:
“Why ST elevation if no infarction?”
Answer:
Myocardial stunning and catecholamine toxicity
VIVA 5: Microvascular Obstruction (MVO)
Examiner:
CMR shows transmural LGE with MVO. Troponin already peaked.
Question:
Which biomarker predicts mortality better now?
Answer:
NT-proBNP.
Why:
Hemodynamic stress and remodeling dominate prognosis.
VIVA 6: Type 2 MI vs Myocarditis
Examiner:
Septic patient, troponin high, echo global hypokinesia, CMR no focal LGE.
Diagnosis?
Type 2 MI with myocardial injury.
Push:
How would myocarditis differ on CMR?
Answer:
- Subepicardial or mid-wall LGE
- Regional edema pattern
VIVA 7: OCT vs CMR Discordance
Examiner:
OCT shows plaque rupture. CMR shows no LGE.
How do you reconcile?
Answer:
Rupture without sustained occlusion → no necrosis yet.
Examiner follow-up:
Management implication?
Answer:
High-risk ACS, needs aggressive antithrombotic therapy despite no infarct.
VIVA 8: Chronic Troponin Elevation
Examiner:
CKD patient with hs-troponin persistently elevated, no delta, IVUS shows stable plaque.
Classification?
Chronic myocardial injury.
Trap:
Do NOT label NSTEMI without delta.
VIVA 9: hs-CRP vs Anatomy
Examiner:
Angiography mild disease. hs-CRP 10 mg/L.
What does hs-CRP tell you?
Answer:
Systemic inflammatory risk, not culprit anatomy.
Push:
Does it guide PCI?
Answer:
No.
VIVA 10: GDF-15 Oral Trap
Examiner:
Two NSTEMI patients, same troponin and EF. One has very high GDF-15.
Who dies first and why?
Answer:
High GDF-15 patient due to frailty, bleeding risk, systemic vulnerability.
VIVA 11: D-dimer Confusion
Examiner:
STEMI with very high D-dimer. What do you do differently?
Answer:
Nothing. Prognostic only.
Trap:
Do NOT escalate anticoagulation based on D-dimer.
VIVA 12: Reinfarction vs Troponin Kinetics
Examiner:
Post-PCI, troponin still rising at 24 h.
Reinfarction?
Answer:
No. Expected kinetics.
Push:
What confirms reinfarction?
Answer:
New rise after fall + clinical/ECG evidence.
VIVA 13: MINOCA
Examiner:
Troponin positive, angiography normal.
First imaging to do?
Answer:
CMR.
Why:
Differentiates myocarditis, Takotsubo, true MI.
VIVA 14: OCT-Guided Therapy
Examiner:
OCT shows erosion, minimal thrombus.
Would you stent?
Answer:
Not necessarily. Consider conservative antithrombotic strategy.
Examiner trap:
“Guideline?”
Answer:
Evidence evolving (EROSION studies), individualized decision.
VIVA 15: Ultimate Integrative Question
Examiner:
Which modality answers WHAT question best?
| Question | Best tool |
|---|---|
| Did myocardium die? | CMR (LGE) |
| Is there active necrosis? | Troponin |
| Is patient hemodynamically stressed? | NT-proBNP |
| Is plaque unstable? | OCT / MPO |
| Who will bleed or die? | GDF-15 |
🔑 FINAL ORAL-VIVA MANTRA
“Biomarkers tell physiology,
Imaging tells anatomy,
CMR tells truth,
Troponin needs context.”