Brugada Syndrome
Brugada Syndrome

| Short Question | Brugada Syndrome |
|---|---|
| What is Brugada Syndrome? | A genetic arrhythmia disorder with characteristic ECG changes and risk of sudden cardiac death. |
| Most common genetic mutation? | SCN5A gene mutation. |
| Typical inheritance pattern? | Autosomal dominant. |
| Typical ECG hallmark? | Coved-type ST-segment elevation in V1–V3. |
| Which ECG lead placement can unmask the pattern? | High intercostal placement of V1–V2. |
| Most common arrhythmia in Brugada Syndrome? | Polymorphic ventricular tachycardia or VF. |
| Drug used for diagnostic challenge? | Ajmaline or flecainide. |
| Which gender is more affected? | Males (about 8:1 male-to-female ratio). |
| Most dangerous complication? | Sudden cardiac death. |
| Definitive treatment for high-risk patients? | Implantable cardioverter-defibrillator (ICD). |
| Common trigger for arrhythmia? | Fever. |
| What should be avoided in Brugada Syndrome? | Sodium channel-blocking drugs unless for diagnosis. |
| Region of heart primarily affected? | Right ventricular outflow tract (RVOT). |
| Type 1 Brugada ECG pattern description? | Coved ST elevation ≥2 mm with inverted T wave in V1–V3. |
| Geographical prevalence? | More common in Southeast Asia and Japan. |
| First described by? | Pedro and Josep Brugada in 1992. |
| Can Brugada be asymptomatic? | Yes, many cases are found incidentally on ECG. |
| Prognosis without treatment in high-risk patients? | Poor, due to high risk of sudden death. |
| Can ablation help? | Epicardial ablation of RVOT substrate may reduce arrhythmia risk. |
| Role of family screening? | Recommended for first-degree relatives. |
| # | Brugada Syndrome |
|---|---|
| 1 | Brugada Syndrome is a genetic disorder affecting cardiac sodium channels. |
| 2 | Most cases are linked to mutations in the SCN5A gene. |
| 3 | Characterized by coved-type ST-segment elevation in V1–V3 on ECG. |
| 4 | Often presents with syncope or sudden cardiac arrest, especially during rest or sleep. |
| 5 | More common in men than women, with higher prevalence in Southeast Asia. |
| 6 | Triggers include fever, certain medications, and electrolyte imbalances. |
| 7 | Type 1 ECG pattern is diagnostic; types 2 and 3 require provocation testing. |
| 8 | Ajmaline or flecainide challenge can unmask concealed Brugada patterns. |
| 9 | Risk stratification is based on symptoms and family history of sudden cardiac death. |
| 10 | ICD implantation is the mainstay of therapy in high-risk patients. |
| 11 | Quinidine can be used in patients who are not ICD candidates or have recurrent ICD shocks. |
| 12 | Catheter ablation may target arrhythmogenic substrate in the RV outflow tract. |
| 13 | Febrile illnesses should be treated aggressively to prevent arrhythmic events. |
| 14 | Certain drugs such as Class I antiarrhythmics, tricyclic antidepressants, and lithium should be avoided. |
| 15 | ECG changes may be intermittent and influenced by autonomic tone. |
| 16 | Family screening is essential for early detection in asymptomatic carriers. |
| 17 | The arrhythmia mechanism often involves phase 2 reentry in the RV epicardium. |
| 18 | Polymorphic ventricular tachycardia or VF are typical terminal rhythms in events. |
| 19 | ECG lead placement modification (high V1–V3) can improve detection. |
| 20 | Brugada phenocopies can mimic ECG changes but are caused by reversible conditions. |
Brugada syndrome, Brugada type 1 ECG, SCN5A mutation, RVOT ablation, ajmaline challenge, flecainide challenge, fever unmasking Brugada, ICD in Brugada, sudden cardiac death risk,,LSI/Supporting Terms: coved ST elevation, V1–V3 leads, sodium channelopathy, polymorphic VT, ventricular fibrillation, quinidine therapy, Brugada phenocopy, high intercostal V1–V2
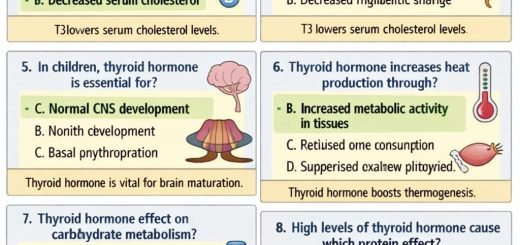
ECG patterns
There are three main types of ECG patterns linked to Brugada syndrome, primarily seen in the right chest leads (V1-V3):
- Type 1 (diagnostic): Features a coved-type ST-segment elevation of 2 mm or more, followed by a negative T wave. This pattern is considered diagnostic.
- Type 2: Shows a saddleback-shaped ST-segment elevation of 2 mm or more at the high take-off, a trough with at least 1 mm of ST elevation, and a positive or biphasic T wave.
- Type 3: Displays either a saddleback or coved-type ST-segment elevation less than 1 mm.
Note: Only the Type 1 ECG pattern confirms Brugada syndrome. Type 2 and Type 3 patterns may require further tests, like a drug challenge, to reveal a Type 1 pattern and confirm the diagnosis.
ECG lead placement
Sometimes, placing the right chest leads (V1-V3) higher on the chest (in the second or third intercostal space) can help reveal a subtle or hidden Brugada ECG pattern, especially Type 1.
Other ECG characteristics
Other ECG findings that may be present but are not specifically diagnostic include:
- Incomplete right bundle branch block (RBBB)
- Prolonged PR interval
- Left axis deviation
- Atrial fibrillation
| No. | Key Point about NOS |
|---|---|
| 1 | Nitric oxide synthase (NOS) converts L-arginine into nitric oxide (NO) and citrulline. |
| 2 | There are three main isoforms: nNOS (NOS1), iNOS (NOS2), eNOS (NOS3). |
| 3 | nNOS is expressed in neurons and skeletal muscle. |
| 4 | eNOS is expressed in endothelial cells, regulating vascular tone. |
| 5 | iNOS is inducible by cytokines and produces large amounts of NO during inflammation. |
| 6 | NO activates soluble guanylyl cyclase, increasing cyclic GMP (cGMP). |
| 7 | nNOS and eNOS are calcium/calmodulin dependent. |
| 8 | iNOS is calcium-independent once induced. |
| 9 | Important cofactors: FAD, FMN, NADPH, Heme, BH₄, Ca²⁺/Calmodulin. |
| 10 | ADMA (asymmetric dimethylarginine) is an endogenous inhibitor of NOS. |
| 11 | eNOS dysfunction is linked to hypertension, diabetes, and atherosclerosis. |
| 12 | Excessive iNOS activation contributes to septic shock and tissue damage. |
| 13 | nNOS regulates synaptic plasticity, learning, and memory. |
| 14 | Gene locations: NOS1 on chromosome 12, NOS2 on 17, NOS3 on 7. |
| 15 | Sildenafil enhances NO effect by preventing cGMP breakdown (PDE-5 inhibition). |
| 16 | Direct NO donors include nitroglycerin and sodium nitroprusside. |
| 17 | BH₄ deficiency reduces NO synthesis, causing endothelial dysfunction. |
| 18 | NO prevents platelet aggregation and leukocyte adhesion to endothelium. |
| 19 | NO is a free radical but physiologically protective at normal levels. |
| 20 | NO imbalance contributes to cardiovascular, neurological, and inflammatory disorders. |